Two new medications have recently entered the limelight of medical weight loss. Championed by celebrities and influencers, everyone is asking their doctor for one or both of these substances to help them lose weight, but are these drugs the right choice for your patients?
The drugs in question are Semaglutide (brand names: Wegovy, Ozempic) & Tirzepatide (brand name: Mounjaro). Both of these drugs are FDA approved medications for Type 2 Diabetes, given as a weekly subQ injection.
- Semaglutide/Tirzepatide Costs
- Where to get Semaglutide/Tirzepatide
- How does Semaglutide/Tirzepatide work?
- Side Effects
- Muscle Wasting
- Long Term Weight Loss
- Natural Alternatives
What does Semaglutide cost?
The average retail (patient) cost of semalutide (Ozempic) and tirezepatide (Mounjaro) is $1,172 for a 4-week supply[depending on dosage] (GoodRx, 2023). While these drugs are approved for treatment of type 2 diabetes, insurance may not cover the cost of these medications for weight loss. Without insurance, semaglutide or tirezepatide could cost a patient over $14,000 per year.
Where to buy Semaglutide
Semaglutide is a patent-protected compound, which means it can only be produced by the pharmaceutical corporations who hold the patent. However, an exception to this restriction is made when for drugs that are considered to be in shortage – any drug which appears on the drug shortage list can be compounded by a 503B compounding pharmacy.
Is compounded semaglutide safe?
503B compounding pharmacies (also known as 503B outsourcing facilities) are different from traditional compounding pharmacies because they must comply with the FDA’s CGMP requirements and are inspected by the FDA on a risk-based schedule to ensure they are meeting these requirements.
“There are greater assurances of quality when drugs are compounded by outsourcing facilities that meet the conditions of section 503B and CGMP requirements” [FDA]
Does semaglutide need to be refrigerated?
Refrigeration requirements may vary depending on the pharmacy and their production methods. Lyophilized (powder) forms of semaglutide do not need to refrigerated until they are reconstituted. After reconstitution refrigerate up to 28 days.
How do they work?
Semaglutide and Tirzepatide work in very similar ways. Both Semaglutide and Tirzepatide are GLP-1 agonists; Tirzepatide is also a GIP agonist.
AGONIST: BIOCHEMISTRY
A substance which initiates a physiological response when combined with a receptor.
GLP-1 (glucagon-like peptide-1) and GIP ( glucose-dependent insulinotropic peptide ) are normally stimulated by the presence of food in the small intestine. GLP-1 and GIP are both known as incretins, which are defined as gut hormones that can increase the amount of insulin released from the pancreas (Stipanuk & Caudill,510). All incretins control insulin and a satiety signal, and therefore have an impact on adiposity and glucose levels.
Do they work for weight loss?
Double-blind studies (published in the New England Journal of Medicine) appear to show positive effects on weight loss from both Semaglutide and Tirzepatide compared to placebo.
SEMAGLUTIDE resulted in weight reductions of 5% (1047 vs 182), 10% (893 vs 69), & 15% (612 vs 28) for more patients than placebo.
“Nausea and diarrhea were the most common adverse events with semaglutide; they were typically transient and mild-to-moderate in severity and subsided with time. More participants in the semaglutide group than in the placebo group discontinued treatment owing to gastrointestinal events (59 [4.5%] vs. 5 [0.8%])”Wilding, J. et al. (2021).
TIRZEPATIDE resulted in weight reductions of 5% (89% vs 35%), & 20% (50% vs 3%) for more patients than placebo.
“The most common adverse events with tirzepatide were gastrointestinal, and most were mild to moderate in severity, occurring primarily during dose escalation. Adverse events caused treatment discontinuation in 4.3%, 7.1%, 6.2%, and 2.6% of participants receiving 5-mg, 10-mg, and 15-mg tirzepatide doses and placebo, respectively.”Jasterboof, A. et al. (2022).
Semaglutide & Tirzepatide Side Effects
Nearly all FDA-approved drugs have side-effects, and semaglutide/tirzepatide are no exception. In fact, studies on semaglutide for weight loss all contain a common phrase:
“Participants in the semaglutide group discontinued treatment owing to gastrointestinal events.”
Semaglutide side effects:
- Nausea
- Vomiting
- Diarrhea
- Abdominal pain
- Constipation
- Heartburn
- Burping
Tirzepatide side effects:
- Nausea
- Vomiting
- Diarrhea
- Loss of appetite
- Stomach pain
- Constipation
- Upset stomach
A new JAMA study on semaglutide found that:
- Almost half (48.6%) of the patients using semaglutide reported experiencing adverse effects such as…
- Nausea
- Vomiting
- GI symptoms
- Which leads to patient attrition and non-compliance.
To relieve these side-effects many physicians and their patients have turned to natural solutions including digestive enzymes, a probiotic, and natural cleansing herbs to promote regularity, which can be found in our SEMAGLUTIDE SUPPORT KIT:

SEMA-ZYME is a special formulation of eight natural digestive enzymes which can prevent bloating. By effectively supplementing digestive enzymes daily, digestive enzymes may promote more complete digestion of carbohydrates, proteins, and fats, as well as better absorption of nutrients, helping to ensure continued good health.
SEMA-BIOTIC is a unique combination of the superior room temperature stable probiotic LactoSpore®, a digestive enzyme blend Digezyme®, and Ginger extract. This combination has been designed to support optimal digestive function, contributing to healthy metabolism and relieving side-effects such as nausea.
SEMA-SMOOTH daily cleanse is a natural cleansing formulation that is mild and fast acting. Contains natural cascara sagrada, rhubarb root, ginger root and aloe curacao for effective results. Ridding the colon of waste and toxins by releasing layers of buildup can lead to feelings of clarity, strength and overall good health. This is a non-habit forming formulation.
Semalgutide & Muscle Wasting
In the STEP 1 study, 1961 adults were randomized to receive semaglutide 2.4 mg once-weekly or matched placebo for 68 weeks. Participants who received the drug lost an average of 10.4kg (23.8%) of fat mass and 6.9kg (13.2%) of lean body mass.
A smaller study known as SUSTAIN 8, researchers followed 53 semaglutide users for 52 weeks and found that during their weight loss they had lost 3.4kg (10.2%) of fat mass and 2.3kg (4.5%) of lean body mass.
In both studies the results were the same: While the semaglutide group had significant weight loss, ~40% of the weight lost was from a decline in lean muscle mass.
According to a spokesperson for Novo Nordisk, the company “did not specifically study the medicine’s impact on muscle mass” in its clinical trials.
Muscle loss can be minimized by increasing cAMP:
In addition to promoting fat burning, cAMP encourages development of lean muscle. Researchers have demonstrated that “cAMP participates in muscle development and regeneration mediated by muscle precursor cells” (Berdeaux & Stewart, 2012).
In the muscles, increasing cAMP results in increased activity of glycolysis, an ATP-producing process that begins with the breakdown of glucose. Studies have found that low glycolytic activity is associated with reduced muscle size (Tixier et al, 2013).
Muscle growth is the result of a complex process that adds more myosin “filaments” to each muscle fiber.
In the liver, cAMP promotes gluconeogenesis, producing glucose from non-carbohydrate sources. This can have an appetite-sparing effect while simultaneously providing fuel for the muscles. Together these effects result in increased lean body mass, decreased fat mass, and a reduced appetite – naturally!
Increasing cAMP without drugs
FASTING: When fasting the body maintains its energy requirements by accessing stored glucose and lipids. One of the mechanisms through which this is accomplished is an increase in cAMP.
FORSKOLIN: Forskolin is a natural compound extracted from the Indian coleus plant (Coleus forskohlii). It exerts its effects by activating the enzyme adenylate cyclase, which stimulates the production of cAMP. Forskolin’s ability to increase cAMP levels has made it a subject of scientific research and has been studied for its potential therapeutic applications in conditions like obesity.

How fast does semaglutide work for weight loss?
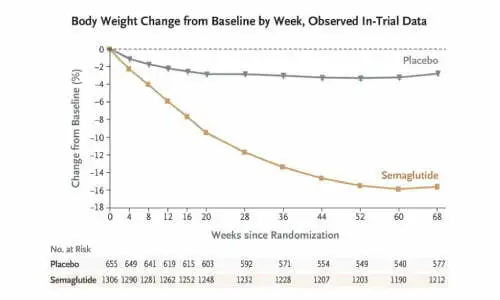
Studies have shown that semaglutide can result in weight loss in as little as 4 weeks, with results peaking at 60-68 weeks. However, studies have also shown that weight rebound can occur just as quickly after discontinuing treatment with semaglutide.
Semaglutide’s Long Term Success in Question
Research published in the journal Diabetes, Obesity and Metabolism indicates that once people stop using the medication semaglutide (better known by the brand names Wegovy and Ozempic), any weight they’ve lost is likely to return.
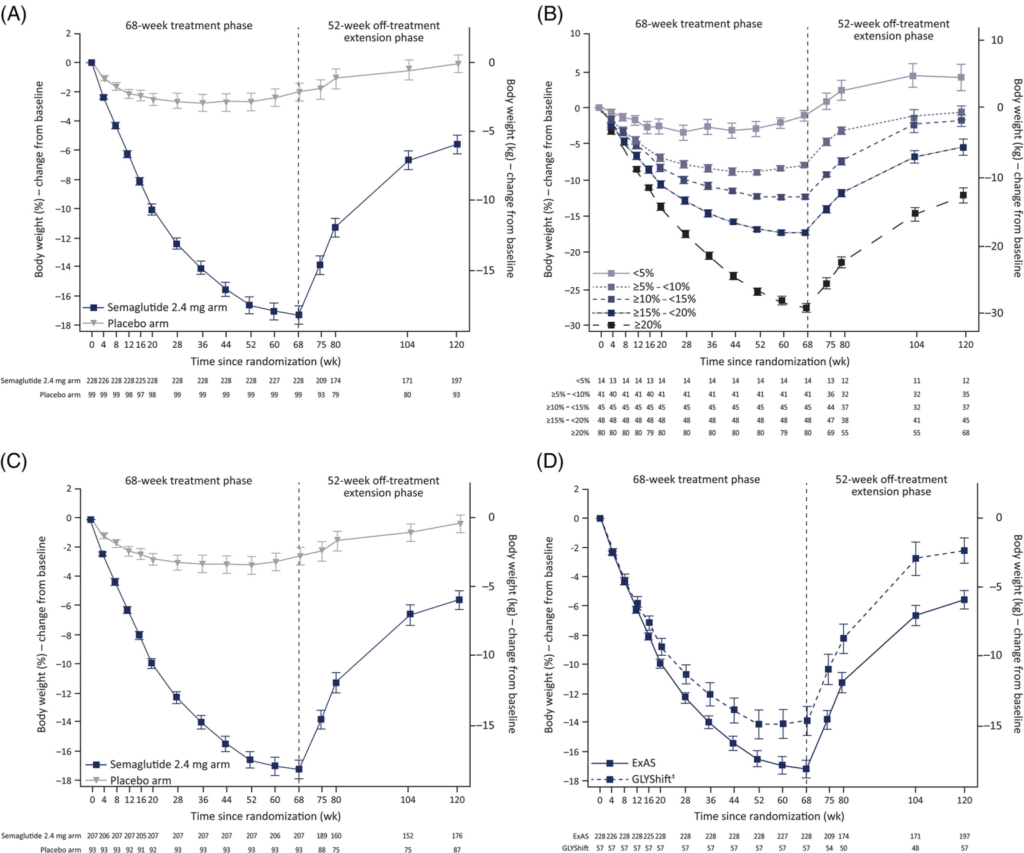
Another study published in April 2022 found that a year after discontinuing semaglutide, people had regained two-thirds of the weight they had lost. Other positive changes they had seen in cardiometabolic risk factors like blood pressure, blood lipids, HbA1c, and C-reactive protein had similarly reversed.
“After withdrawal of semaglutide and structured lifestyle intervention, participants regained a mean of two‐thirds of their prior weight loss in the 1‐year off‐treatment extension phase; weight regain continued until the end of follow‐up “ (2)
A double-blind study published in JAMA had patients use semaglutide for 20 weeks, then compared the effects of continuing semaglutide vs switching to a placebo. The researchers found that the patients who switched to placebo gradually regained the weight, while the semaglutide group did not. (4)
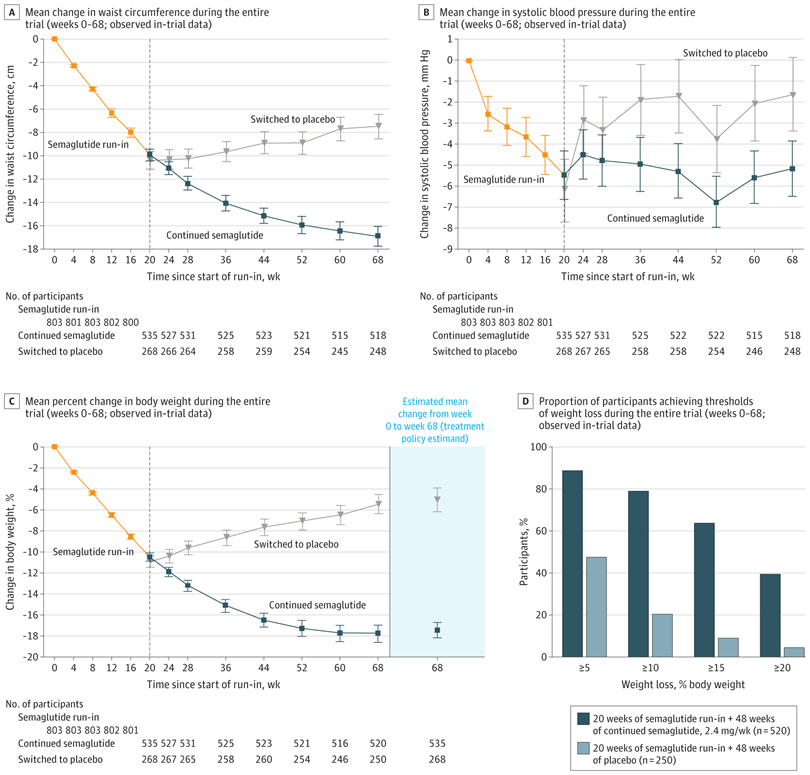
Why do patients regain weight after semaglutide?
Weight rebound occurs because the drug does not cure the underlying issues that led to weight gain in the first place.
This is illustrated by the following image which shows that in patients with prediabetes, blood glucose levels normalized during treatment with semaglutide, but returned to prediabetes levels after discontinuing semaglutide, suggesting the underlying issue which caused prediabetes had returned.
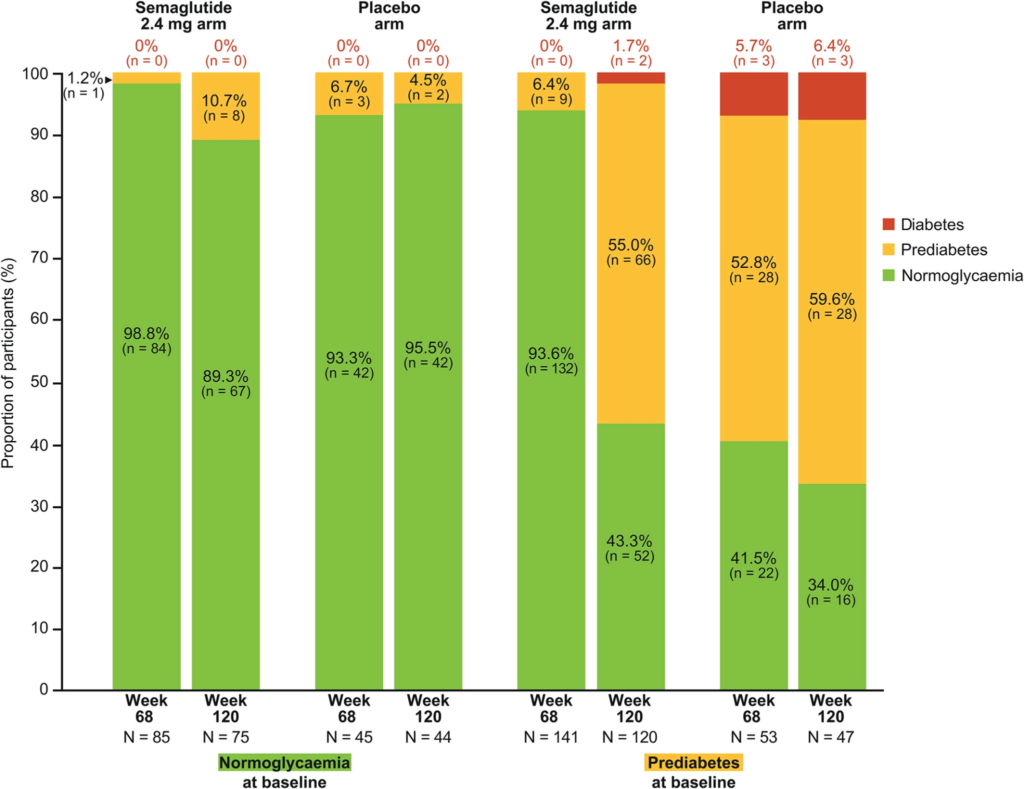
This finding is significant because it implies that the underlying issue is INSULIN RESISTANCE.
Insulin is released in response to glucose in the blood stream where it facilitates the removal of glucose from the blood. After glucose is removed from the blood, it can be burned for energy or stored as fat. Insulin accomplishes this by communicating with the cells mitochondria, but when cells have an impaired response to these communications we consider them to be insulin resistant. Left untreated, insulin resistance can lead to prediabetes and type 2 diabetes.
This conclusion would be consistent with other studies which show higher levels of post-weight loss rebound in patients with elevated markers of insulin resistance, demonstrating that insulin sensitive individuals maintain weight loss better than those with insulin resistance.
“Insulin resistance may play a negative role in maintaining weight loss ”
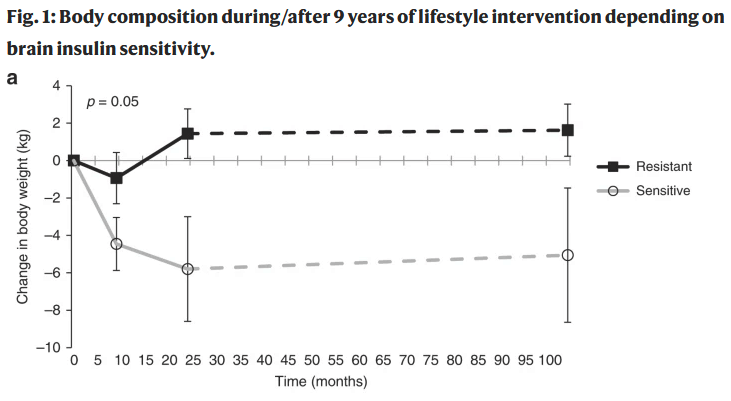
When starting a weight loss regiment, patients with higher levels of insulin sensitivity achieved a greater reduction in body weight and total adipose tissue. “By contrast, insulin-resistant individuals showed a slight weight loss in the first 9 months of the program, and already regained body weight as well as total and visceral adipose tissue during the subsequent months of lifestyle intervention” […] In the long-term follow-up, 9 years after the lifestyle intervention, baseline insulin sensitivity was associated with less regain in body weight” (1). Patients with higher insulin sensitivity also saw the greatest reduction of total dietary intake during the study period.
Another study which followed 40 individuals, grouped based on insulin sensitivity at the start of the program, found that after 12 months of a weight loss program the group with the highest level of insulin sensitivity had maintained weight loss significantly better than the insulin-resistant group. (3)
EDITORS NOTE: While there have been some claims of semaglutide improving insulin sensitivity, studies find that any positive effects on insulin resistance from semaglutide are due to the weight loss rather than the action of semaglutide directly
Natural Options for Long Term Success
As mentioned previously, the primary mechanism of action of these drugs is to increase activation of GLP-1. GLP-1 is normally activated by the presence of food in the gut, but there are other substances which can influence this incretin such as Momordica charantia and Gymnema Sylvestre.
Momordica charantia (Bitter Gourd) has demonstrated hypoglycemic activity in a number of clinical studies, and is believed to act through a number of pathways, including acting on GLP-1. “Our data provide evidence that BG stimulates GLP-1 secretion which contributes, at least in part, to the antidiabetic activity of BG through an incretin effect” (Huang et al, 2013).
Gymnema Sylvestre has been used in ayurvedic medicine for over 2,000 years where it has been nicknamed the “sugar destroyer”. Part of Gymnema’s sugar destroying ability comes from GLP-1. A 2020 study concluded that Gymnema Sylvestre “has significant antihyperglycemic activity through stimulation of GLP-1 release and its enhanced proglucagon expression in ileum followed by Insulin Receptor (IR) expression in pancreas” (Kilari et al, 2020).
These natural GLP-1 agonists are complimented by nutrients which have a positive impact on insulin function and glucose management such as chromium & Vanadyl Sulfate.
Chromium: Chromium is an essential mineral that helps to enhance the action of insulin in the body. It does this by binding to insulin receptors on cells, which allows insulin to better transport glucose from the bloodstream into cells. This can help to improve metabolism and energy levels.
Vanadyl Sulfate: Vanadyl Sulfate is a compound that has been shown to improve insulin sensitivity and regulate blood sugar levels. It works by mimicking the effects of insulin in the body, which can help to reduce insulin resistance and improve glucose uptake by cells.
DNS contains therapeutic levels of these key ingredients which have been shown to:
- Increase Glucose Utilization (increases metabolism of glucose)
- Stimulate Glucose Uptake
- Improve Insulin Sensitivity
- Improve Glucose Tolerance
- Reduce Sugar Cravings


